Introduction
Loss of tolerance toward several self-antigens suggests an underlying genetic predisposition. A previous study (1) conducted in children with Evans syndrome revealed a high prevalence of pathogenic mutations in genes involved in primary immunodeficiencies. Whether these findings are restricted to children is unknown. In this study, we assessed the prevalence of constitutional genetic variants in a cohort of patients with adult onset Evans syndrome.
Patients and methods
We consecutively included from November 2021 to June 2023 patients that had been diagnosed with adult onset (≥18 years) Evans syndrome (association of immune thrombocytopenia (ITP), warm autoimmune hemolytic anemia (wAIHA) and/or autoimmune neutropenia (AIN) according to international guidelines (2,3)), and identified in France throughout the network of the national reference center for adult' autoimmune cytopenia. We excluded patients with a systemic lupus (according to Systemic Lupus International Collaborating Clinics criteria (4)), and patients with indolent B cell malignancies at Evans syndrome onset. Clinical and biological data were retrospectively collected using a standardized form. Patients underwent whole-exome sequencing after written and informed consent was obtained. Gene variants already published and/or with in vitro confirmed consequences were considered as genetic diagnoses. Clinical interpretation of variants was performed according to the ACMG guidelines (5). A patient was classified as having a genetic diagnosis if carryinghemizygous/heterozygous pathogenic or likely pathogenic variant (for X-linked or autosomal dominant conditions), or 2 pathogenic or likely pathogenic variants in the same gene (for autosomal recessive conditions).
Results
We identified 80 patients (40% female, median age 31 years at Evans syndrome diagnosis [range 18-76]) with adult-onset Evans syndrome from 19 centers. Median age at first cytopenia was 28 years [range 1-73], and 13 patients (16%) were under 18 years at first cytopenia. 78 patients (96%) had ITP (chronic in 87%), 59 (73%) had wAIHA (with at least one relapse in 58%), and 38 (47%) had AIN (chronic in 74%). Median delay between first and second cytopenia was 18 months (range 0 - 329) and 28 patients (35%) had synchronous onset of cytopenia.
Twenty-four patients (30%) had associated immune manifestation (involving endocrine system in 9%, lung in 9%, gastrointestinal tract in 6%, skin in 5%, central nervous system in 4%) and 28% had antinuclear antibody titers ≥ 1/160. Lymphoproliferation was found in 56%, including lymphadenopathy in 41% and splenomegaly in 35%. Nine patients (11%) had lymphoma or cancer history. Twenty patients (25%) had humoral immune defect, including 13 (16%) with a common variable immunodeficiency (CVID)-like phenotype.
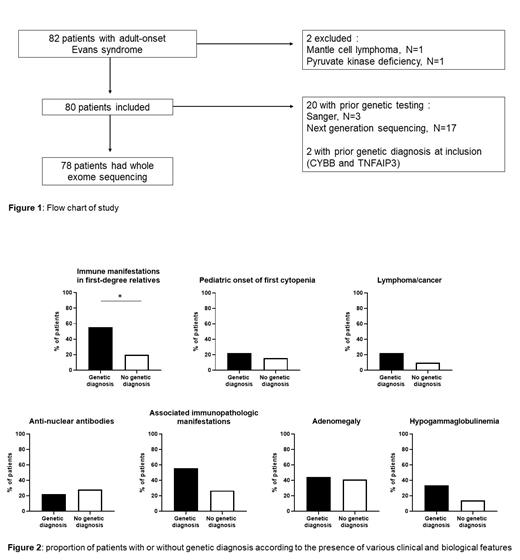
In the whole cohort, 9 patients (11.25%) had a genetic diagnosis that could be linked to Evans syndrome, with heterozygous variants in CTLA4, STAT3, NFKB1, CYBB, TNFAIP3, DDX41, MYH9 and homozygous variant in PIK3CG. Twenty (25%) patients had genetic testing prior to this study (targeted Sanger sequencing [N=3], or NGS panel [N=17]), resulting in 2 genetic diagnoses ( CYBB and TNFAIP3). A family history of immune manifestations in first-degree relatives was found in 56% of the patients with genetic diagnosis versus 20% of patients without (P=0.0308). However, the proportion of patients with CVID-like phenotype, associated immune disorder, lymphoproliferation, cancer/lymphoma, positive antinuclear antibodies, or pediatric onset of first cytopenia was not significantly increased in patients with genetic diagnosis.
Discussion
As expected, the proportion of genetic diagnoses in patients with adult onset Evans syndrome was lower that what was previously described in children, but led to a significant impact on clinical management in some patients. We did not find clinical or biological features associated with a genetic diagnosis, suggesting that genetic testing should be considered in every adult patient diagnosed with Evans syndrome in the absence of any obvious underlying cause or condition.
References
(1) Hadjadj J et al., Blood 2019; 134: 9-21.
(2) Provan D et al., Blood Adv 2019; 3: 3780-817.
(3) Jäger U et al., Blood Rev 2020; 41: 100648.
(4) Petri M et al., Arthritis Rheum 2012; 64: 2677-86.
(5) Richards S et al., Genet Med 2015; 17: 405-24.
Disclosures
Crickx:Novartis: Honoraria; Sanofi: Honoraria; UCB: Honoraria. Cheminant:Abbvie: Research Funding; Amgen: Honoraria; Innate Pharma: Research Funding; AstraZeneca: Other: Travel accomodations and Meeting inscription. Godeau:Novartis: Honoraria; Amgen: Honoraria; Grifols: Honoraria; Sobi: Honoraria. Galicier:EUSAPHARMA: Consultancy; AMGEN: Consultancy. Viallard:EUSAPHARMA: Consultancy. Audia:Amgen: Honoraria; Novartis: Honoraria; Sanofi: Honoraria. Oksenhendler:CSL Behring: Consultancy; EusaPharma: Consultancy. Hermine:AB science: Consultancy, Current equity holder in publicly-traded company, Current holder of stock options in a privately-held company, Research Funding; Inatherys: Consultancy, Current equity holder in private company, Research Funding; BMS, Roche, Blueprint, Alexion: Research Funding. Mahevas:Amgen: Honoraria; Novartis: Honoraria; Sanofi: Research Funding. Michel:Sobi: Consultancy; Alexion: Consultancy; UCB: Honoraria; argenx: Honoraria; Sanofi: Consultancy; Novartis: Consultancy.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal